An article by Y. Cao, Y. Liang, L. Zhang, S. Osuna, A.-L. M. Hoyt, A. L. Briseno*, and K. N. Houk* is featured in J. Am. Chem. Soc.
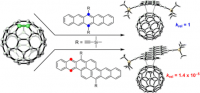
The Houk (UCLA) and Briseno groups investigated the Diels–Alder reactions of pentacene (PT) and bistetracene (BT), with the [6,6] double bond of [60]fullerene. Reaction barriers and free energies were obtained via DFT calculations to assess the effects of frameworks and substituent groups on the DA reactivity and product stability. Calculations indicate that BT is about 5 orders of magnitude less reactive than PT in reactions with [60]fullerene. This accounts for the observed much higher stability of BT than PT when mixed with [60]fullerene.