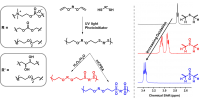
A communication by Joel M. Sarapas and Gregory N. Tew published in Angewandte Chemie International Edition.
The thiol-ene reaction has been widely used in the field of polymer science for decades, primarily as a network generation and polymer functionalization tool. In this communication, we demonstrate the utility of this reaction to generate functional, linear polymers capable of undergo selective redox-tuning via the thioether inherent to this method. We target materials containing a main-chain carbonate, main-chain zwitterion, or side-chain diol, though the possible breadth of this platform is nearly infinite. Due to the air, water, and functional group tolerance of this reaction, the thiol-ene step-growth polymerization is a strong addition to the functional polymer synthetic toolbox.